
- Written by ZILA GmbH
- font size decrease font size increase font size
CO2 in industrial processes: Personal and plant protection by reliable carbon dioxide monitoring
CO2 is a natural component of our atmosphere. The total amount of CO2 in dry air is about 0.038 % or 380 ppm. This low concentration is not dangerous for humans. However, CO2 is emitted in high concentrations during combustion and fermentation processes and is also used in many industrial processes due to its characteristics.
Gases are substances which, in their natural form, have a gaseous state of aggregation under standard conditions of temperature and pressure. If the temperature drops, the gas firstly changes into a liquid state and becomes a solid substance at even lower temperatures. In this way, CO2 becomes dry ice, since dry ice is frozen carbon dioxide.
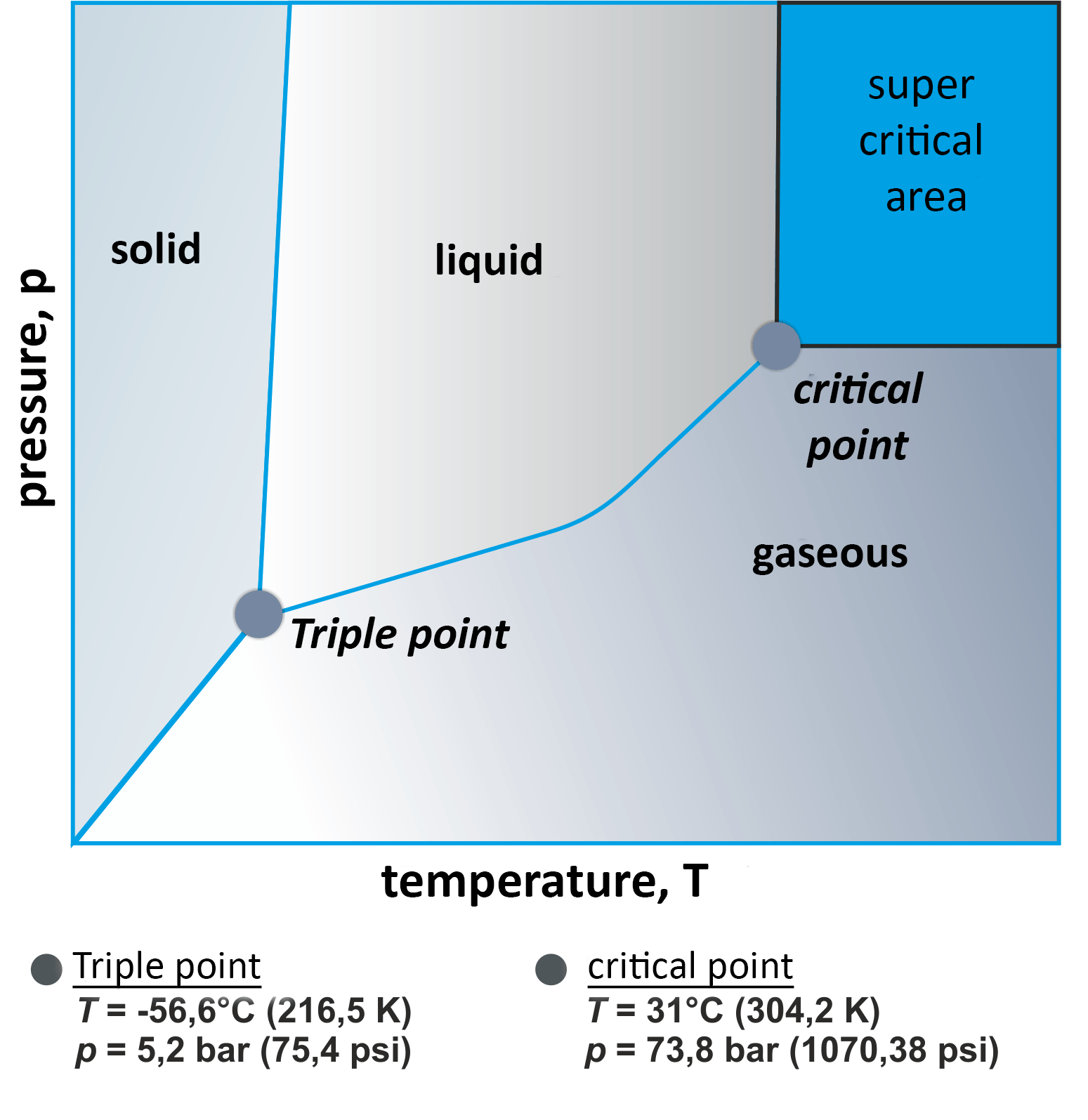
Carbon dioxide can turn into dry ice when it is in gaseous, liquid or solid state. It is colourless, odourless and heavier than air when gaseous. The state of the substance can be modified by changing the pressure conditions. For instance, carbon dioxide can be made to reach the supercritical or overcritical state at 73.8 bar and 31°C. In this state, CO2 is a perfect solvent and separating agent.
Another interesting aspect is the triple point of CO2 at 5.2 bar and -56.6°C. At this point, the substance's different states of aggregation can appear simultaneously. By cleverly changing the parameters for pressure and temperature, CO2 can be used as an excellent and comparatively inexpensive refrigerant.
Application areas of CO2
Because of these favourable characteristics, carbon dioxide is necessary as an industrial gas in many sectors. For example, CO2 is an excellent natural refrigerant in refrigeration plants and is also suitable as a test gas. On the one hand, the ripening process of fruit and vegetables in greenhouses can be accelerated by controlled CO2 fertilisation, and on the other hand, it can be slowed down during storage and transport. In dispensing systems as well as in beverage production, CO2 is added to beverages as an additive.
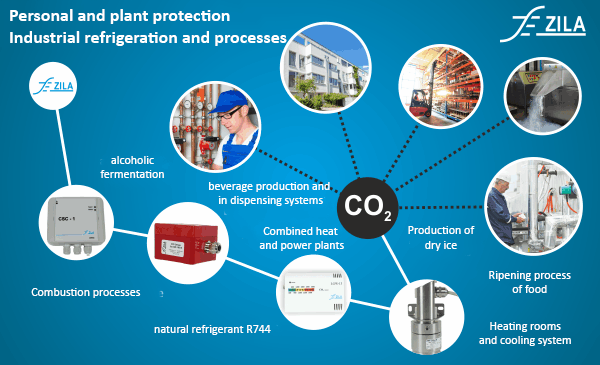
CO2 monitoring for personal and plant protection
The increasing use of CO2 demonstrates the qualities of carbon dioxide. However, caution is required when dealing with carbon dioxide.
The gas CO2 cannot be perceived by humans. Health hazards and damage to machines and plants can arise in many ways. Incorrect operation, leakages and accidents can result in CO2 being released uncontrollably. If the oxygen content in the air we breathe in drops, this can lead to serious consequences in living and working areas. Starting at a concentration of 8 % (80,000ppm [parts per million]), CO2 can cause unconsciousness and death within 30-60 minutes. Higher concentrations even accelerate this process.
Therefore, exact and permanent monitoring and early detection are necessary.
CO2 gas detection with sensors
A tested and approved method for measuring the CO2 concentration is the optical measuring method NDIR. This involves penetrating the air mixture with infrared radiation and measuring the absorption of a specific wavelength in the infrared spectrum. Sensors for CO2 detection using this method are generally very durable and almost maintenance-free.
ZILA GmbH can look back on more than 25 years of experience and competence with handling liquid and gaseous media. Our product solutions range from compact and precise sensors for living areas (air quality) up to the sturdiest sensors for industrial applications. You can choose from measuring ranges between 0...100vol%. In addition, we integrate our sensor technology into devices for controlling fans and other actuators.
Sensors for CO2 detection
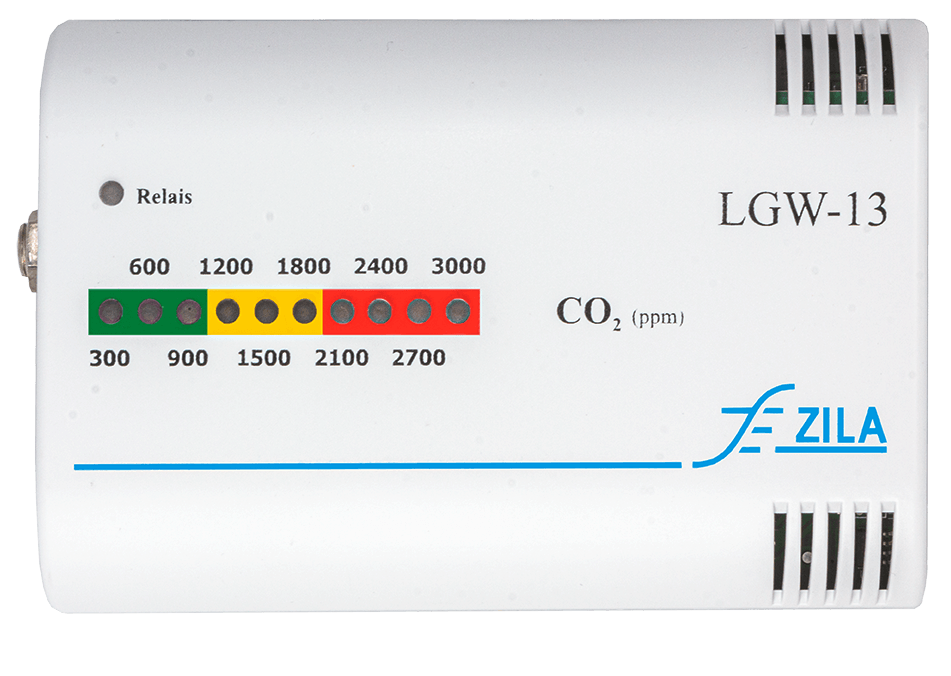
Carbon dioxide air quality guard with switching function and warning signal transmitter

Climate control KST-20 Vento/CO2
Climate control for automatic air quality measurement and room dehumidification

Industrial CO2 sensor ZMF-100-IR
Robust carbon dioxide sensor for industrial applications with optical measuring principle NDIR
More information
Site map: Gas detection



